The mechanisms of regeneration and their evolution
Most animals are able to repair wounds and many can regenerate extensively, re-growing organs or even entire body plans from small fragments. A full understanding of the mechanisms, i.e., how wounding results in this “whole-body” regeneration, is lacking. Further, it is unknown whether regeneration mechanisms are conserved or independently evolved across diverse animal species. Thus, our lab seeks to: 1) identify cellular and genetic mechanisms for regeneration and stem cells, and 2) understand how regeneration has evolved by comparing mechanisms across distantly-related animals.
The model system
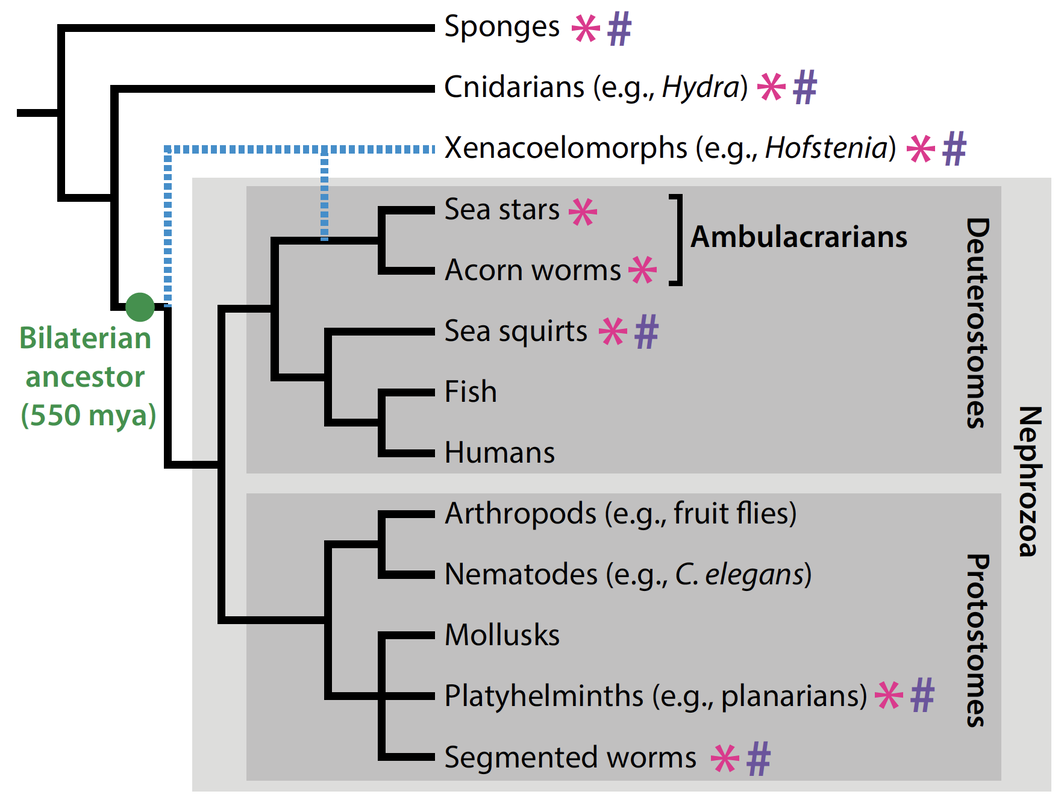
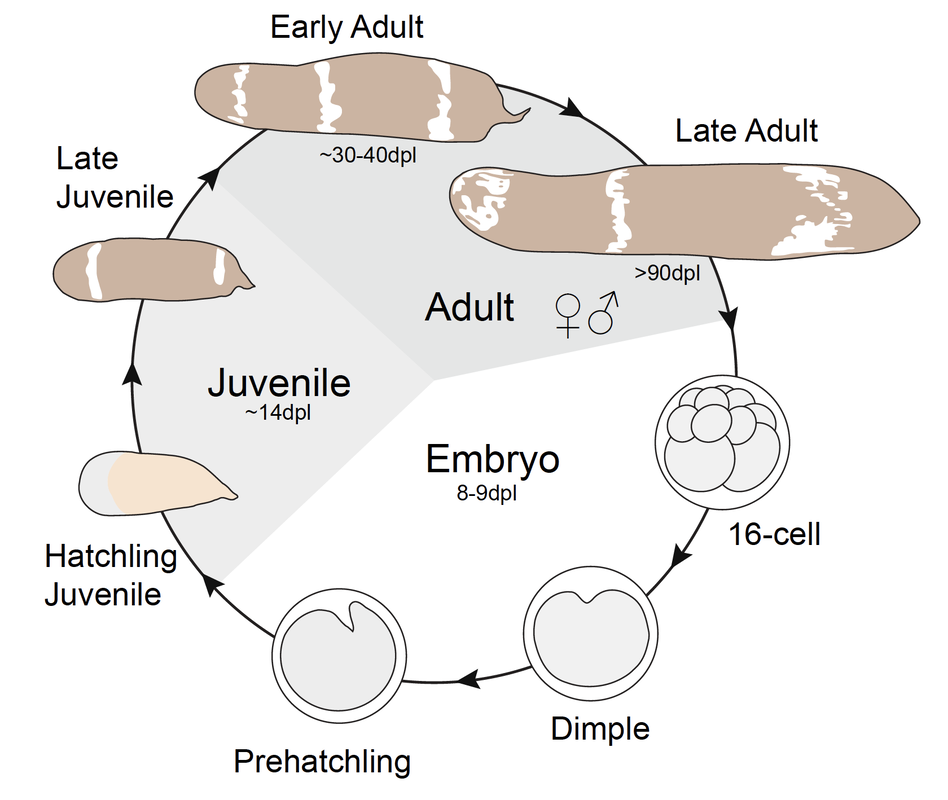
Our work focuses on a new model system that enables deep mechanistic insights into the process of regeneration and facilitates comparisons of distantly related animal lineages. Our model, the acoel worm, Hofstenia miamia, a.k.a. the three-banded panther worm, belongs to a lineage (Phylum: Xenacoelomorpha) that is distantly-related to traditional regenerative model invertebrates such as planarians (550 mya) and hydra (650 mya). Hofstenia presents many advantages as a model regenerative species, with many genomic/transcriptomic resources and tools for functional studies including stable transgenics and CRISPR-Cas9 genome editing.
|
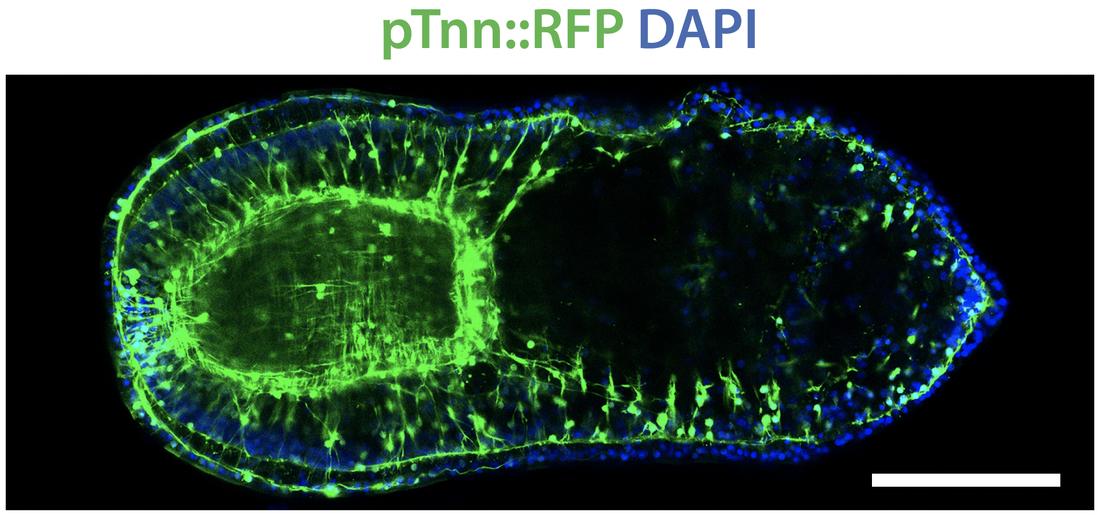
Transgenic embryo showing muscle development.
|
The questions and approaches
|
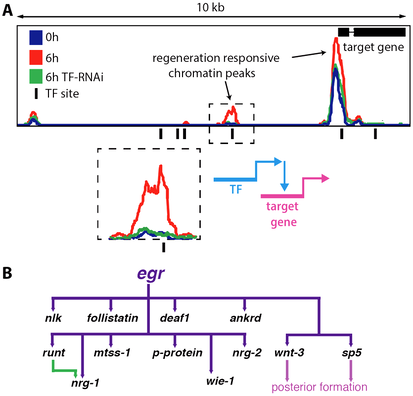
Regeneration: Whole-body regeneration in Hofstenia hinges upon several crucial events including signaling from the wound site (i.e., the wound response), proliferation of stem cells, and patterning of the newly formed cells to correctly integrate new tissue into the existing animal body plan. Seeking to understand these major aspects of regeneration, we utilize functional epigenomics approaches in Hofstenia to build gene regulatory networks for regeneration.
|
|
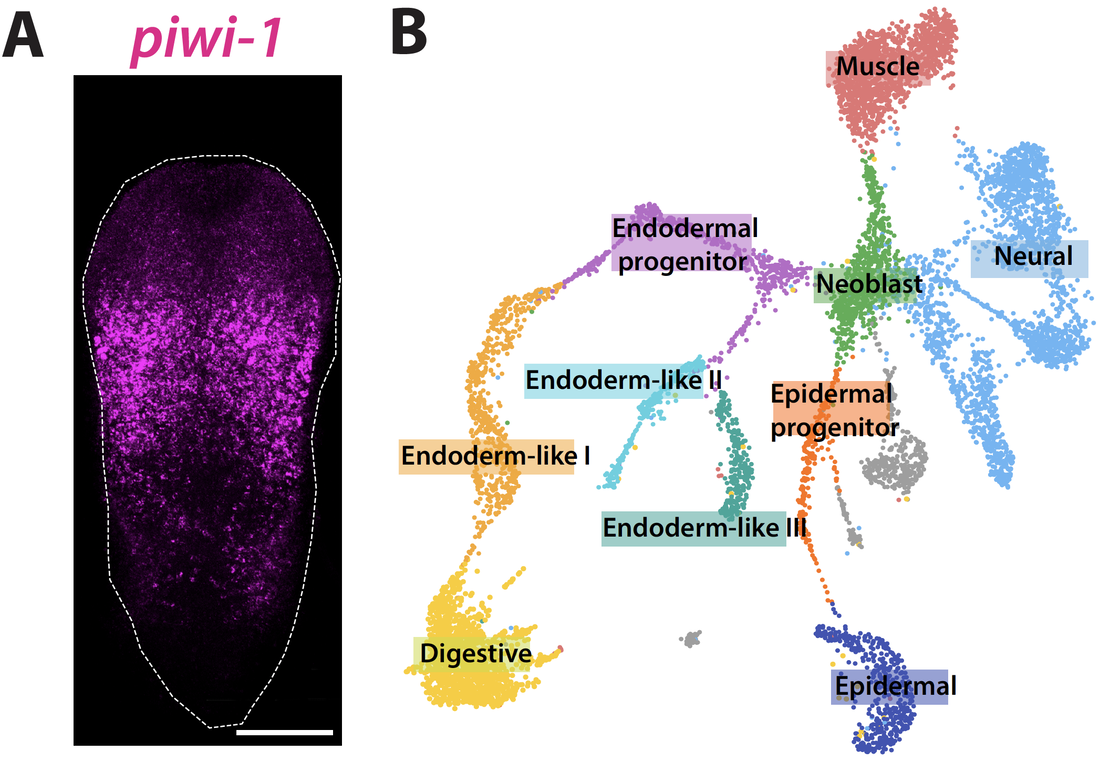
Stem Cells: Hofstenia miamia harbors a large population of adult pluripotent stem cells called "neoblasts". We utilize single-cell RNA-sequencing data to understand how neoblasts work during regeneration and how they are made during embryogenesis.
|
|
New questions: Hofstenia is an excellent laboratory system, which has opened up many new research directions in the lab. Examples include studies that 1) compare regeneration and development, 2) examine novel cell biology, such as the close contacts that muscle fibers make with other cells, and 3) assess the structure and function of the brain.
|